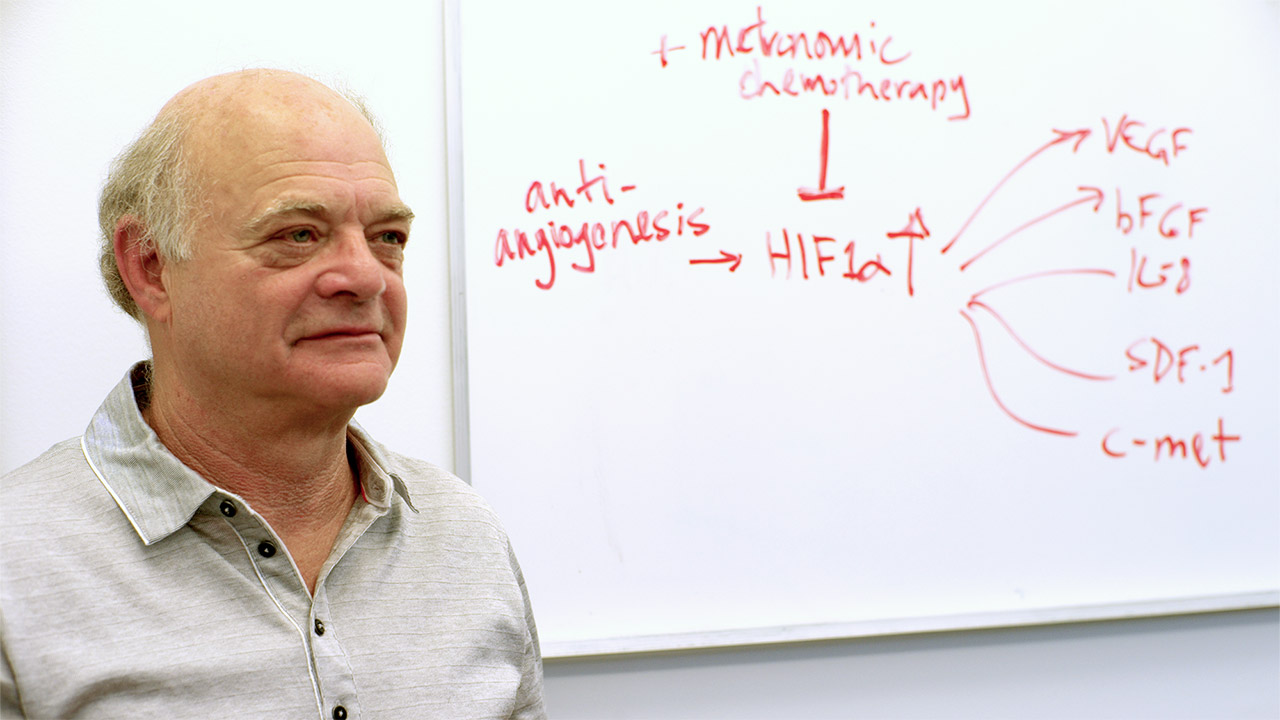Chemotherapy concept pioneered at Sunnybrook Research Institute highlighted in The Lancet
Sunnybrook Research Institute Senior Scientist Dr. Robert Kerbel shares insight on progress made in the field of metronomic chemotherapy in an invited editorial commentary “Adjuvant metronomic chemotherapy for locoregionally advanced nasopharyngeal carcinoma,” recently published in The Lancet.
Unlike traditional chemotherapy regimens, where patients are often administered the maximum tolerated dose (MTD) and then given a 2-3 week break to recover from the treatment’s side effects, 'metronomic chemotherapy' utilizes low doses of chemotherapy given continuously over a much longer period of time, and without long breaks because of its reduced toxicity. This makes it potentially attractive for use in what are known as adjuvant or maintenance treatments where patients have previously received a conventional therapy, often using MTD chemotherapy, but are at risk of their disease recurring, and cannot continue a toxic MTD chemotherapy protocol.
In the commentary, Dr. Kerbel, a pioneer of metronomic chemotherapy, explains why a phase III clinical trial published in the same issue of The Lancet, may be a significant milestone.
“Over 20 years ago, my lab and another at Harvard led by the late Judah Folkman, came up with the idea that there may be circumstances in which chemotherapy drugs can be given at much lower doses over extended periods of time on a much more frequent, even daily, schedule, usually resulting in much less toxicity, but without necessarily compromising efficacy,'' says Dr. Kerbel. “This regimen was later termed metronomic chemotherapy because of the frequent fast “beats” of giving the drug, like a metronome.” It is thought to work by several mechanisms, depending on the drug used, which include inhibiting new blood vessel formation in tumours ('antiangiogenesis') or stimulating the body’s immune response to tumour cells as well as inhibiting tumour cells directly.
Dr. Kerbel has devoted much of the last two decades to researching the therapy, with numerous preclinical studies that helped pave the way for in-human clinical trials. His body of work in this area encompasses a total of 64 papers since 2000. Over the last few years, metronomic chemotherapy has been successfully evaluated as a maintenance therapy or adjuvant therapy in a number of pivotal randomized controlled phase III clinical trials, undertaken in Europe, India and China.
Given that most of the drugs used for metronomic chemotherapy are oral and off-patent and can be taken at home in pill form, they are significantly less expensive and more convenient than MTD chemotherapy regimens given by intravenous infusions. “Importantly, this makes them a more accessible option for patients in low- and middle-income countries,” says Dr. Kerbel.
In The Lancet, Dr. Kerbel and French pediatric oncologist Dr. Nicolas Andre, highlight the affordability and convenience of metronomic chemotherapy, while discussing the aforementioned phase III clinical trial in The Lancet that evaluated a metronomic chemotherapy regimen as an adjuvant/maintenance therapy in patients at risk for relapse of nasopharyngeal carcinoma after they had received conventional concurrent radiation/chemotherapy that successfully controlled their locoregional disease. The multi-centre trial, led by investigators at Sun Yat-sen University Cancer Center in Guangzhou, China, showed a survival benefit in patients who received the low-dose continuous metronomic regimen using a drug called capecitabine, every day for one year at home compared to those who received no treatment, the current accepted ‘standard therapy’. In their commentary, Dr. Kerbel and Dr. Andre also argue for a change of mindset in the pharmaceutical industry about how metronomic chemotherapy using off-patent drugs might be utilized, especially for long-term adjuvant or maintenance treatments, either alone or in combination with other types of newer on-patent drugs such as immunotherapy agents for use in high-income countries.
“This trial and a number of others recently evaluating metronomic chemotherapy where the results have been positive have been very gratifying, personally,” says Dr. Kerbel. “It represents the culmination of more than two decades of work, which also involved an uphill struggle to demonstrate that the conceptually counterintuitive treatment may have merit. Whether this will result in a change in North America with respect to attitudes about metronomic chemotherapy remains to be seen, but with more research we’re hopeful we can continue to demonstrate its significant potential.”