Participate
Who can participate?
Patients of the Sunnybrook sleep clinic with newly diagnosed sleep apnea of sufficient severity may be eligible to participate in the Brain Changes in Sleep Apnea Study. Eligible participants must be newly diagnosed with sufficiently severe sleep apnea but not yet receiving any treatment for it. Eligible participants must also be planning to start CPAP treatment for their sleep apnea and be able to undergo a brain MRI safely (see FAQs).
How do I participate?
If you would like to participate in the BCSAS, then please contact the study coordinator by phone (416-480-5143) or email (sleepapneabrain@sunnybrook.ca), or through the Contact Us section of the website. He can answer any questions or concerns that you may have about the study.
What is involved?
If you participate in the Brain Changes in Sleep Apnea Study, then you will complete a series of recordings using wearable devices in the comfort of your home. In addition, you will come to Sunnybrook for a half-day of tests.
In the comfort of your home, you will do the following:
- Wear a wrist watch-like accelerometer (Figure A) 24 hours a day for eight days. This tells us when you are sleeping and how well you are sleeping.
- Wear a simple portable device (Figure B) to measure your breathing and oxygen levels in your sleep for one night.
- Wear a portable blood pressure monitor (Figure C) 24 hours a day for one day. This tells us your blood pressure levels throughout the day.
- Collect your urine for 24 hours in a special jug. We will provide the jug for you.
Instructions on how to use the study devices at home will be provided for you.

Figure A (GeneActiv)

Figure B (WatchPAT)
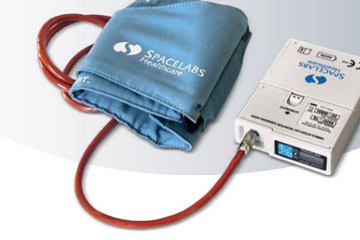
Figure C (SpaceLabs 90217 blood pressure monitor)
During a half-day visit to Sunnybrook, you will do the following:
- Provide a blood sample.
- Have a few tests of memory and concentration.
- Have a brief test of the stiffness of your blood vessels.
- Have an MRI of your brain. The MRI will take about one hour.
You will then repeat these measurements after four months of treatment with a CPAP machine as prescribed by your usual sleep physician.
How will this study benefit me?
You will not benefit directly from participating in this study. The physical measurements collected by the Brain Changes in Sleep Apnea Study are only for research purposes. This means that the physical measurements are not diagnostic and are not designed to replace routine clinical care. That being said, at the end of your participation in this study, we will mail you summaries of the sleep, blood pressure and breathing measurements you recorded at home.
Although you will not benefit directly from participating in this study, your participation may further our understanding of the impact of sleep apnea on brain function and brain diseases like stroke, dementia, and Alzheimer’s disease. The results of this study may lead to new treatments to prevent or reverse the impact of sleep apnea on brain health.
For participants
Video instructions for WatchPAT sleep monitor
View consent form (PDF) (also available in Word format)
View study instruction sheets (PDF) (also available in Word format)
Contact information
Email: sleepapneabrain@sunnybrook.ca
Telephone: 416-480-5143
Address: Sunnybrook Health Sciences Centre, 2075 Bayview Ave., M1 600, Toronto, ON M4N 3M5


